THEN NOW NEXT
Homing In on HCM
By Jill Stefancin
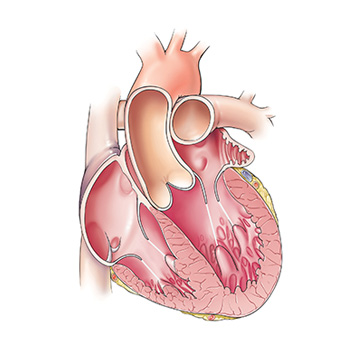
HCM can result in thickening of the septum, the muscular wall that separates the left and right sides of the heart. | Illustration: Joe Pangrace
THEN
First described at an autopsy in 1958, hypertrophic cardiomyopathy (HCM) is a complex type of heart disease that causes thickening of the heart muscle, left ventricular stiffness and mitral valve changes. Symptoms include chest pain, palpitations, shortness of breath, fatigue and fainting. The exact cause of HCM is often unknown or attributed to genetics, high blood pressure or aging. It’s the most common cause of sudden cardiac death in people under age 30. By the mid-1960s, medications such as betablockers, calcium channel blockers and antiarrhythmics were prescribed to treat the symptoms. Other treatment options included heart surgery or alcohol septal ablation, therapies used to reduce thickening of the septum, the wall separating the right and left sides of the heart.

Dr. Milind Desai | Photo: Marty Carrick
NOW
A Cleveland Clinicled study — funded in part with generous philanthropic support — showed that an experimental drug, mavacamten (Camzyos®), significantly reduced the need for invasive procedures. Based largely on the clinical trial’s results, the FDA approved the drug in April 2022 for treatment of adults with symptomatic obstructive HCM. The first-ever precision medication for treatment of HCM, mavacamten works by reducing excessive contraction of the heart muscle, making it work more efficiently. It also reduces the stiffness of the heart muscle. “This disease has been barely recognized for about 60-odd years, so the evolution of diagnostic techniques, the evolution of invasive techniques and now the evolution of precision medications in this disease is huge if you take the spectrum of time we are talking about,” says the principal investigator of the study, Milind Desai, MD, MBA, Director of the Hypertrophic Cardiomyopathy Center in the Sydell and Arnold Miller Family Heart, Vascular & Thoracic Institute at Cleveland Clinic. Follow-up of the study’s participants will continue at least 128 weeks, and one-year data will be available later in 2023.
NEXT
Cleveland Clinic is also one of a small number of centers involved in early studies of a gene therapy approach to HCM. The strategy targets a protein deficiency mutation and the most prevalent genetic form of HCM. The approach involves an investigational virus-based gene therapy designed to address the underlying cause of HCM. “The virus is administered as a single injection to stimulate protein formation and theoretically result in normal myocardial thickness,” Dr. Desai explains, noting that preclinical studies have shown successful mass regression. “If it’s ultimately shown to be safe and effective in humans, it could prove to be a one-time solution for a fairly common form of HCM.”
